Clinical Guidelines for the Medical Management of Hepatitis C
E. Treatment of Hepatitis C
The primary goal of HCV therapy is to achieve a SVR, defined as an undetectable HCV RNA 6 months after stopping antiviral therapy. Secondary goals of antiviral therapy include improvements in histology, quality of life and prevention of hepatocellular carcinoma. Antiviral therapy is approved by the Food and Drug Administration (FDA) for patients with persistently abnormal liver enzymes, detectable HCV RNA and an abnormal liver biopsy. Recent data have shown that patients with normal liver enzymes, detectable HCV RNA and an abnormal liver biopsy respond to therapy at similar rates as those with abnormal liver enzymes.55
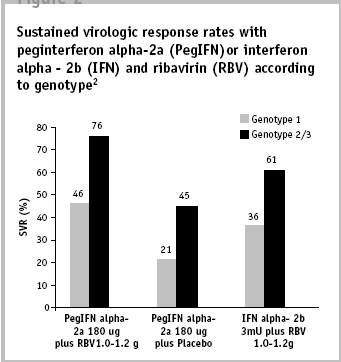
The efficacy of HCV treatment has improved over the past decade. Initial treatment consisting of interferon alpha has been replaced by pegylated interferon and now by combination therapy using pegylated interferon and ribavirin. Efficacy varies depending on multiple factors especially viral genotype, but achieving sustained viral suppression in 50% of patients can be expected (see Figure 2).
Figure 2.
1. Patient Evaluation and Treatment
Recommendations
Treatment should be considered for all patients with detectable HCV RNA and an abnormal liver biopsy, regardless of the presence or absence of liver enzyme elevation.
Prior to making a decision regarding treatment, patients should be evaluated with HCV RNA, HCV genotype, liver enzymes (ALT), and liver biopsy, unless contraindicated. The decision to initiate antiviral therapy should be made based upon the willingness of the patient to undergo therapy, ability to regularly attend appointments, and agreement to use contraception to prevent pregnancy. The decision to initiate antiviral therapy should be made on an individualized basis that considers severity of liver disease, co-morbid conditions, the potential for serious side effects and the likelihood of response.
Patients with HCV infection on methadone maintenance therapy should not be considered ineligible for treatment.
The treatment of the actively using injection drug user is not contraindicated and may be appropriate under some circumstances. Patients with a history of well-controlled psychiatric disorders may be excellent candidates for antiviral therapy and should be under the care of a qualified mental health professional.
Treatment of HIV/HCV co-infected patients should be offered with pegylated interferon and ribavirin, unless contraindicated. Patients co-infected with HIV/HCV should be managed by experts in both viruses. The basic tenets of HCV management should not change, but the provider must be prepared for possible hepatotoxicity and drug-drug interactions. Further recommendations for the HIV/HCV co-infected patient are provided at: CRITERIA FOR THE MEDICAL CARE OF ADULTS WITH HIV INFECTION
All patients with CHC infection are candidates for antiviral therapy. These patients are defined by detectable serum HCV RNA and an abnormal liver biopsy consistent with chronic liver disease. Treatment is recommended for patients with significant inflammation or fibrosis.23
There are relatively few contraindications to antiviral therapy although the decision to initiate therapy should be made after ensuring that the patient understands the risks and benefits of pegylated interferon and ribavirin. Current absolute contraindications to combination therapy include a known hypersensitivity to pegylated interferon and/or ribavirin, autoimmune hepatitis, decompensated liver disease, pregnant women, men whose female partners are pregnant and patients with hemoglobinopathies.56,57 Many patients with hepatitis C will also have underlying mental illness such as depression. Uncontrolled psychiatric illness and suicidal ideations or attempts are contraindications to antiviral therapy. Patients with remote histories of suicidal ideation or attempt warrant further evaluation to assess suitability for treatment. On the other hand, patients whose psychiatric disorders are under control and who are regularly followed by mental health providers are often excellent candidates for antiviral therapy.
IDU is the most common mode for acquisition of HCV infection. Patients with a history of injection drug use who are no longer using recreational injection drugs are treated in the guidelines as noted above. Methadone use has not been shown to adversely affect SVR rates or interfere with patient adherence to medication regimens. Patients enrolled in methadone maintenance programs should be considered for antiviral therapy.58
The treatment of actively injecting drug users is controversial and raises concerns related to adherence to therapy and the potential for re-infection. Patients actively using injection drugs should be offered drug counseling and psychiatric support services. Like all patients, treatment of the actively injecting drug using person should be based upon the willingness of the patient to undergo therapy, ability to regularly attend appointments for close monitoring, and agreement to use of contraception to prevent pregnancy.
2. Environmental Assessment and Support
Environmental support is an important part of patient assessment because treatment may be given for up to one year, and the adverse effects of treatment may incapacitate patients. A patient's living situation and household income should be addressed prior to treating treatment. Homelessness may be a significant problem, and the need for a support network for such patients should be assessed and arranged before the treatment. In addition, most formulations of pegylated interferon now require refrigeration. Family meetings can be helpful to prepare family members for side effects of treatment. Neuro-psychiatric side effects such as irritability and hostility can strain relationships if unexpected. These issues can be assessed with the collaboration of social services. Family and friends may need to help with activities of daily living including transportation to medical appointments. Home health nurses and case managers may be helpful in providing support at home.
3. Initiating Treatment
Recommendations
Prior to treatment, patients should have a baseline complete blood count (CBC), chemistry evaluations, serum creatinine, thyroid function tests, pregnancy tests in women, HIV testing, contraceptive counseling for men and women, and screening for depression.
Prior to initiating treatment, patients should be informed of the possible side effects of therapy to allow them to anticipate and manage with these side effects.
The treatment of choice for patients with chronic hepatitis C infection is combination pegylated interferon and ribavirin. Patients infected with genotype 1 or 4 should be treated for 48 weeks with combination pegylated interferon and ribavirin. The ribavirin dose should be 1000 mg a day in patients <75 kg and 1200 mg a day in patients >75 kg.
Patients infected with genotype 2 or 3 should be treated for 24 weeks with combination pegylated interferon and ribavirin. The ribavirin dose should be 800 mg a day.
Several treatments are licensed in the U.S. for the treatment of CHC. These agents include interferon alpha-2a, interferon alpha-2b, interferon alpha con-1, and interferon alpha-2b in combination with ribavirin; pegylated interferon alpha-2b alone and in combination with ribavirin; and interferon alpha-2b and pegylated interferon alpha-2a alone and in combination with ribavirin. Data from multiple clinical trials clearly supports the use of pegylated interferon in combination with ribavirin.
Pegylated interferon has been a major advance in the treatment of CHC. The concept behind the pegylation of interferon is to produce a molecule which maintains longer lasting therapeutic concentrations by optimizing both absorption and distribution while decreasing the rate of clearance and deceasing proteolysis. This is accomplished by the addition of a polyethylene glycol molecule [PEG] to standard interferon by way of a covalent bond. This PEG molecule is non-toxic polymer that is readily excreted in the urine. The PEG molecule can be either linear or branched. Larger PEG molecules produce greater reductions in renal clearance and provide more subcutaneous absorption.
Two pegylated molecules are currently being used in the U.S. Pegylated interferon alfa-2b (Peg-Intron; Schering-Plough) is a linear 12 KD molecule, and pegylated interferon alfa-2a (Pegasys; Roche) is a 40KD branched chain molecule. These products are both manufactured using recombinant DNA technology in an Escherichia coli system. Both products have dose related maximum concentrations. Pegylated interferon alfa-2a is given as a fixed dose whereas pegylated interferon alfa-2b is dosed according to patient weight.
One study compared once weekly pegylated interferon alfa-2a with standard interferon alfa-2a three times a week for 48 weeks in previously untreated patients with hepatitis C.59 The SVR rate in the pegylated interferon group was 39% compared to a 19% response rate in the standard interferon group. The SVR rate of genotype 1 patients receiving peginterferon alfa-2a was 28%. The frequency and severity of adverse events was similar in both groups. Pretreatment factors that were associated with a sustained virologic response in this study, in order of significance, include genotype other than type 1, ALT quotient greater than three, HCV RNA level less than two million copies (Cobas Amplicor HCV-PCR version 2; Roche), body surface area less than 2 meters, lack of bridging fibrosis or cirrhosis, and age less than 40 years. Side effects were less with the group treated with pegylated interferon alfa-2a than the group that received standard interferon therapy.
The highest SVR rates in previously untreated patients with chronic hepatitis C have been reported with combination pegylated interferon and ribavirin. In one study of 1530 patients, a 54% SVR was reported in patients treated with pegylated interferon alfa-2b plus ribavirin.60 This was compared to a 47% SVR rate in patients treated with three times a week standard interferon plus ribavirin. The response rates to pegylated interferon alfa-2b plus ribavirin for genotypes 1 and non-1 were 42% and 82%, respectively.2 In a retrospective analysis of the data, the authors report that patients receiving more than 10.6 mg/kg of ribavirin had higher sustained response rates, regardless of treatment group. Another study showed that patients with genotypes other than type 1, regardless of ribavirin dose and treatment duration, had a sustained reponse of 80%, leading to the recommendation that non-type 1 patients can use a ribavirin dose of only 800 mg and can discontinue treatment after 24 weeks.61
Side effects between the groups receiving pegylated interferon alfa-2b and standard interferon were similar although there was significantly more fever, weight loss, nausea and injection site reactions in the group receiving pegylated interferon alfa-2b. Several studies have documented a decreased sustained viral response rates in African-American patients infected with hepatitis C when compared to Caucasians and Asian-Americans. One prospective study evaluating the SVR rates of African-Americans and whites receiving pegylated interferon alfa-2a plus ribavirin for 48 weeks, reported a response of 26% for blacks compared to 39% for whites.62
4. Monitoring While on Treatment
Recommendations
Patients who do not achieve virologic suppression or a 2-log decrease in HCV RNA at 12 weeks may have therapy discontinued, although factors such as degree of fibrosis and tolerability of therapy should be considered.
Patients should have a CBC and chemistry evaluations 2 weeks after initiation of treatment to assess for potential toxicities. CBC, chemistry evaluations, and pregnancy tests in women should be done routinely at each follow-up visit and not less often then every 4-6 weeks during treatment.
Patients who achieve an end-of-treatment virological response should have HCV RNA testing performed 24 weeks after stopping treatment to evaluate for a SVR.
Erythropoetin alfa and granulocyte colony stimulating factor (G-CSF) may be used to treat anemia and neutropenia, respectively, in order to maintain the patient on full medication doses.
Providers should reference the full discussion of side effects of hepatitis C treatment in Appendix A.
One analysis of pegylated interferon alfa-2b plus ribavirin showed that genotype 1 patients who did not achieve either viral eradication or a drop in baseline HCV RNA by more than 2 log at 12 weeks of therapy had <1% chance of achieving a SVR. Patients who do not achieve this "early viral response" can have therapy discontinued. This action is both cost effective and can improve patient quality of life.63 The key factor in achieving a sustained viral response with pegylated interferon and ribavirin appears to be the patient's ability to adhere to the treatment regimen. Adherence is directly related to side effects and tolerability. Better understanding of the toxicities and side effects of combination therapies and their management should lead to better outcomes. Common side effects of pegylated interferon plus ribavirin therapy include the development of flu-like symptoms, fatigue, alopecia, rash, cough, insomnia, anorexia, thyroid disease, injection-site reactions, vision disorders, anemia, neutropenia, and thrombocytopenia. Rarely, colitis, pancreatitis, and severe pulmonary disease have been observed on alfa-interferon and ribavirin therapy.56,57
Ribavirin may cause birth defects and/or death to the exposed fetus. Extreme care must be taken to avoid pregnancy in female patients and in female partners of male patients taking pegylated interferon plus ribavirin. Ribavirin therapy should not be started unless a report of a negative pregnancy test has been obtained immediately prior to initiation of therapy. Women of child-bearing potential and men must use two forms of effective contraception during treatment and for at least six months after treatment has concluded. Monthly pregnancy tests must be performed routinely throughout the treatment and follow-up phases.64
Pegylated interferon and ribavirin have been found to be safe and effective in HCV mono-infection and in co-infection with HIV.2,61 Safety and efficacy has not been established in patients who have received liver or other organ transplants, in patients who have failed other alpha interferon treatments and in patients under the age of 18.56,57
Neutropenia is commonly seen with pegylated interferon alfa-2b, and 18% of those receiving pegylated interferon at 1.5 ug/kg required a dose reduction due to significant neutropenia. Anemia secondary to ribavirin was also common. Ribavirin dose reduction was seen in 9% of patients treated with pegylated interferon 1.5 ug/kg in combination with 800 mg of ribavirin, and in 13% of those treated with standard interferon and 1000-1200 mg of ribavirin.
5. Re-treatment of Patients Previously Treated for Hepatitis C
Recommendations
Re-treatment of inadequately treated patients is recommended with a combination of pegylated interferon and ribavirin.
Re-treatment of non-responders or relapsers to antiviral therapies other than a combination of pegylated interferon and ribavirin should be strongly considered.
Re-treatment of Patients Who Failed to Respond to Previous Therapies
A large segment of patients with hepatitis C fall into the category of those who did not respond previous to therapy. A careful history must be obtained in these patients to determine if re-treatment should be considered. Patients who did not respond to therapy fall into three general categories: (1) those who were inadequately or inappropriately treated initially, (2) non-responders and (3) relapsers. In addition, patients may fit into any of these three categories after treatments with interferon monotherapy, three-times-weekly interferon plus ribavirin, or pegylated interferon plus ribavirin.
Re-treatment of Inadequately or Inappropriately Treated Patients
Inadequately treated patients are those who either received less than the recommended doses of interferon or ribavirin or were treated for a shorter duration of therapy to make an appropriate assessment as to their response. It is important that all treated patients are encouraged to remain on the baseline dosages of interferon or ribavirin unless untoward effects necessitate dose reduction for a minimum of 12 weeks in order for an assessment of early viral response to be obtained. Inadequately treated patients should be considered for re-treatment with pegylated interferon and ribavirin if no contraindications are noted.
Re-treatment of Non-responders
In patients previously treated with interferon monotherapy, approximately 25-40% may achieve a SVR when treated with pegylated interferon and ribavirin.66,67 Approximately 10% of patients who did not respond to three-times-weekly interferon plus ribavirin will have a SVR with pegylated interferon and ribavirin. Factors associated with a response to retreatment include non-genotype 1 infection, lower baseline HCV RNA levels, less fibrosis on liver biopsy and non-African-American race.66,67 There currently are no published data on the re-treatment of patients who fail to respond to treatment with pegylated interferon and ribavirin.
Re-treatment of Relapsers
Relapse is defined as the reappearance of serum HCV RNA in a patient with previously undetectable HCV RNA at the end of antiviral therapy. Relapse following interferon monotherapy is more common than relapse following combination interferon and ribavirin therapy or combination pegylated interferon and ribavirin therapy.
Large studies show that often relapsers following monotherapy with interferon alone respond favorably to standard interferon plus ribavirin. It seems reasonable to presume that such relapsers will respond to combination pegylated interferon plus ribavirin. The growing problem facing physicians today is how to approach the patient who relapses following combination interferon plus ribavirin or combination pegylated interferon plus ribavirin therapy. Unfortunately, there are limited data currently available to address this issue.
6. Treatment of HCV-Infected Children
Recommendations
Diagnostic evaluation for the presence and severity of HCV infection, including liver biopsy, should be performed in children as in adults.
Therapy with standard interferon and ribavirin may be offered to children aged 3-17 years if given under the care of experienced physicians.
Antiviral therapy should not be administered to children under the age of three.
Children infected with HCV are less likely to manifest symptoms and are more likely to have normal or minimally abnormal liver tests compared with adults. They generally have a slower rate of progression to advanced liver disease. However, there are multiple factors that support treatment of HCV infection in children. These factors include the anticipated long duration of infection after early acquisition, relatively good tolerance of antiviral medications, and avoidance of social stigmatization. Nevertheless, careful selection of appropriate candidates for therapy is important. If a contraindication to current therapeutic agents is present, treatment should be withheld until this has resolved or until new agents are available. Children without contraindications to the medications used for hepatitis C should undergo liver biopsy to determine the presence and degree of fibrosis. In the absence of fibrosis, treatment may be deferred. If any degree of hepatic fibrosis is present, antiviral therapy for HCV should be considered. At present, in the U.S., the only therapy approved for children by the FDA is a combination of interferon alfa-2b and ribavirin. The results of several studies using interferon monotherapy or combination therapy with standard interferon and ribavirin indicate that SVR rates are as good or better than those achieved in adults. Children generally tolerate interferon therapy better than adults; in small numbers of children reported so far dose-dependent hemolytic anemia has been less severe than in adults.68 Although safety data have not yet been developed, pegylated interferon in combination with ribavirin offers improved efficacy and should be considered in adolescents older than 16 years of age who are post-pubertal, or in younger children in the context of clinical trials. Multicenter trials are currently underway to determine the safety and effectiveness of other forms of therapy for HCV infection in children.68 The safety and pharmacokinetics of hepatitis C therapies have not been determined for children younger than 3 years of age.
7. Treatment of Individuals with Acute Hepatitis C Infection
Recommendations
Although there are no controlled trials recommending treatment of acute HCV infection, the use of pegylated interferon monotherapy may prevent the development of CHC infection, although the duration of therapy in still unknown.
There are insufficient data to recommend the use of ribavirin in the acute setting.
Therapy should be deferred until 12 weeks after exposure, to allow for spontaneous clearance to occur, thus avoiding therapy.
The acute phase of HCV infection is seen as a window of opportunity during which the establishment of chronic hepatitis C and its associated morbidity may be prevented.22 A meta-analysis of trials of various interferon alfa monotherapy regimens showed an average SVR rate of 42%, although the quality of these trials was variable.69
Acute HCV infection is seldom seen in clinical practice. Therefore, there is a paucity of well-designed, randomized controlled trials for the treatment of this patient.70 One study reported that intensive treatment of acute hepatitis C with interferon alfa-2b resulted in a 98% SVR. Although there is no standard therapy for the treatment of acute hepatitis C, this study strongly suggests that treatment with interferon monotherapy at higher than standard doses is highly effective in eradicating acute infection.71 Two more recent studies have reported on the effectiveness of pegylated interferon monotherapy for acute hepatitis C.72,73 These studies support initiating therapy after an initial waiting period of approximately 12 weeks after exposure to allow for spontaneous clearance to occur, thus avoiding therapy altogether.