Polio Provider Information
Overview
NYSDOH continues to provide the guidance, resources, and infrastructure needed for healthcare providers in New York to have the information to care for New Yorkers with suspected or confirmed poliovirus infection. Please see the latest guidance.
How can healthcare providers help prevent polio?
- The on-time administration of polio immunizations is critical to keep New Yorkers protected against paralytic polio disease, especially for those in Rockland County, Orange County, and Sullivan County, as well as New York City and Nassau County.
- NYSDOH urges all healthcare providers, especially pediatricians, to ensure their patients are up to date with their polio vaccine schedule.
- Healthcare providers should consider polio in the differential diagnosis of patients with sudden onset of limb weakness, especially with a recent history of fever and/or gastrointestinal illness.
- Providers should especially be on alert for these symptoms in unvaccinated individuals, those currently at increased risk of community transmission, or those with recent international travel or exposures to international travelers.
- Improving vaccination coverage for polio and other vaccine preventable diseases is critical to the public health of all New Yorkers. Providers should take time in every primary care visit, for both adults and children, to ensure that they are up to date with the recommended vaccines for their age.
- Providers should order and stock IPV so they can provide polio vaccination for their patients who are unvaccinated and/or request polio vaccination.
Vaccination
All individuals – children and adults – who are unvaccinated or incompletely vaccinated against polio should be vaccinated with inactivated polio vaccine (IPV) according to the Advisory Committee on Immunization Practices (ACIP) routine and catch-up schedules.
ACIP currently recommends:
- Children who have not started their polio vaccine series or who are delayed in getting all recommended IPV doses should start as soon as possible or finish their series by following the recommended catch-up schedule.
- Adults who are known or suspected to be unvaccinated or incompletely vaccinated against polio should complete a primary vaccination series with IPV.
- Adults who have received a primary series or trivalent oral polio vaccine (tOPV) or IPV in any combination and who are at increased risk of poliovirus exposure may receive another dose of IPV. Available data do not indicate the need for more than a single lifetime booster dose with IPV for adults. Adults at high risk might include:
- Individuals traveling to a country where polio is epidemic or endemic, in accordance with CDC guidance for travelers.
- Laboratory and healthcare workers who handle specimens that might contain polioviruses
- Healthcare providers or other caregivers who have close contact with patients in a community with a polio outbreak
- Other adults who are identified by public health authorities as being part of a group or population at increased risk of exposure because of an outbreak. In New York State, this may include:
- Individuals who collect or work with wastewater specimens for poliovirus testing, especially in areas where poliovirus is circulating.
- Healthcare workers who work in areas with community transmission of poliovirus and who could care for patients with poliovirus (e.g., urgent care, emergency department, neurology, pediatrics).
- Childcare or pre-K providers who work in areas with community transmission of poliovirus and provide diapering or toileting care or assistance.
- Individuals who will or might have exposure to a person known or suspected to be infected with poliovirus, such as household members and other close contacts of a case or suspect case who provide care.
Areas considered to have community transmission of poliovirus include those where poliovirus has been repeatedly detected in wastewater. At this time, that includes Rockland, Orange, and Sullivan Counties, and Brooklyn and Queens within New York City. At this time, booster doses are not recommended for individuals traveling to the New York City metropolitan area, including Rockland, Orange, or Sullivan Counties, merely because of their travel status.
Specimen Collection Guidance
Poliovirus is most likely to be detected in stool specimens, though it may also be detected in pharyngeal swab specimens; identification is less likely in blood or cerebrospinal fluid (CSF). To increase the probability of detecting poliovirus, clinicians should collect stool specimens from patients with illness compatible with suspected paralytic or non-paralytic polio (e.g., unimmunized patients with weakness, paralysis, meningitis, or non-specific viral symptoms), and as early in the course of disease as possible, ideally within 14 days of symptom onset. See the algorithm below for more information.
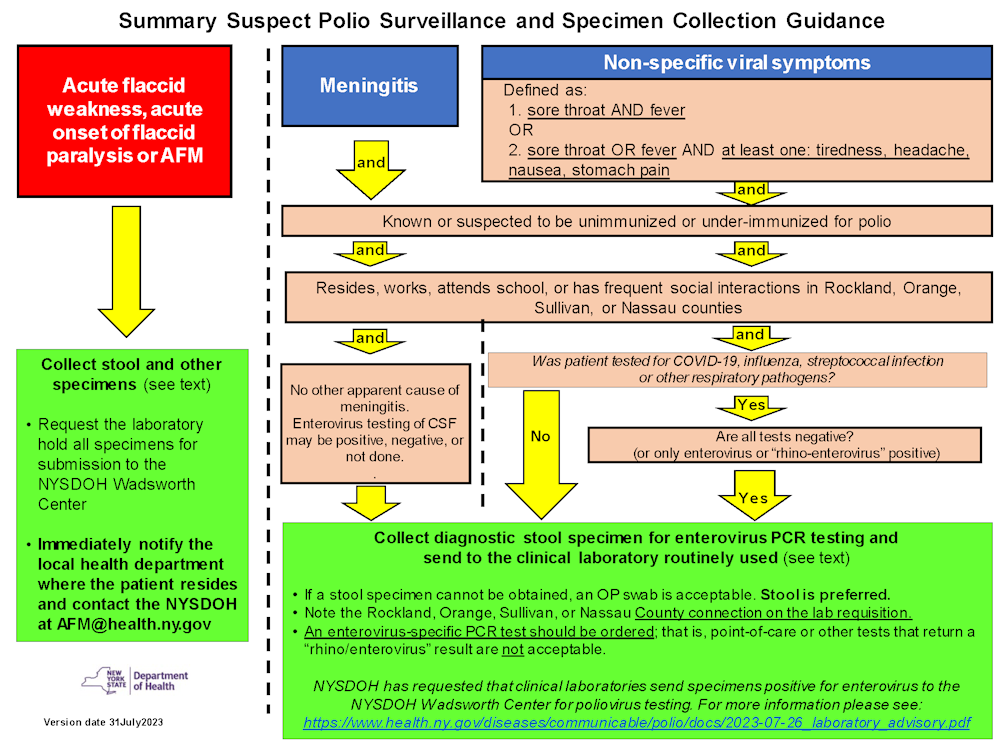
- Flowchart Alt Text Available Here
- Summary Suspect Polio Surveillance and Specimen Collection Guidance (PDF)
Specimen Submission Information
Poliovirus testing is available at NYSDOH Wadsworth Center. Patients with suspected paralytic or non-paralytic polio, AFM, acute onset of flaccid paralysis, or acute flaccid weakness without other known etiology should have a diagnostic stool specimen collected for enterovirus PCR (see above and below). If a stool specimen cannot be obtained, other specimens are acceptable.
Specimens should be collected as follows:
- Two stool specimens (quarter-sized amounts each in a sterile wide-mouth container with no additives) ideally collected separately 24 hours apart. Stool specimens should be kept cold (i.e., refrigerated or frozen). Stool is the highest-sensitivity specimen type for poliovirus and should be a priority for collection.
- Oropharyngeal (OP) swab in viral transport media
- Nasopharyngeal (NP) swab in viral transport media
- Cerebrospinal fluid (CSF) (2-3 cc, if available, in sterile collection tube without additives, frozen promptly to -70/80°C and shipped on dry ice)
- Serum (acute and convalescent), collected prior to treatment with IVIG, (2-3 cc in red or tiger-top tube)
Laboratories performing enterovirus-specific PCR assays should forward positive specimens from unvaccinated or under-vaccinated persons from affected counties (per the requisition form) to the Wadsworth Center as outlined above and in the UPDATED LABORATORY ADVISORY: Submission of Positive Specimens and Isolates from Enterovirus Specific PCR Assays.
A shipping manifest from an electronically submitted Remote Order OR an Infectious Disease Requisition form requesting enterovirus sequencing should accompany all specimens sent to Wadsworth, noting:
- symptoms
- polio immunization history
- whether the individual resides, works, or attends school in southeastern New York (i.e., Rockland, Orange, Sullivan, or Nassau counties).
Specimens should be stored refrigerated and shipped on frozen gel packs. Other routine pathogen-specific testing should continue at hospital and clinical laboratories as indicated. Laboratories should send these specimens immediately and not hold specimens for batched shipments.
Questions
- Clinicians can contact the NYSDOH during business hours at 518-473-4439 or AFM@health.ny.gov or 1-866-881-2809 evenings, weekends, and holidays.
- Clinicians located in New York City can contact the NYCDOHMH Healthcare provider access line at 1-866-692-3641.
- Questions regarding submission of specimens to Wadsworth Center can be directed to wcid@health.ny.gov.
- Questions pertaining to enrollment in the Clinical Laboratory Information Management (CLIMS) for access to remote ordering and access to electronic test reports can be directed to climsoutreach@health.ny.gov.
- For questions regarding immunization, clinicians can call the NYSDOH Bureau of Immunization at 518-473-4437 or email immunize@health.ny.gov.
Provider Resources
- July 31, 2023, Health Advisory (PDF)
- Summary Suspect Polio Surveillance and Specimen Collection Guidance (PDF)
- July 26, 2023, Laboratory Advisory (PDF)
- October 25, 2022, Provider Webinar
- Declaration of an Imminent Threat to Public Health for Polio Virus (PDF)
- September 28, 2022, Health Advisory (PDF)
- Standing Order for IPV Template Letter (DOCX)
- August 19, 2022, Health Advisory (PDF)
- August 4, 2022, Health Advisory (PDF)
- July 22, 2022, Health Advisory (PDF)
- Polio: For Healthcare Providers | CDC
- Polio Vaccination: For Healthcare Providers | CDC
- Report for polio detection in United States - GPEI (polioeradication.org)
- ACIP Polio Vaccine Recommendations | CDC