42 CFR 438 MMC Service Authorization and Appeals -
MMC/HIV SNP/HARP Q&A
- Q&As also available in Portable Document Format (PDF)
Hope Goldhaber, Division of Health Plan Contracting and Oversight
Office of Health Insurance Programs
December 15, 2017; REV January 31, 2018
Welcome
- This presentation is for mainstream Medicaid managed care, HIV SNP and HARP products
- Goals for today:
- Review the most frequently asked questions regarding model notices and the new service authorizations and appeals processes
- Answer additional questions not previously addressed
Reminders
- This webinar is being recorded
- PLEASE:
- DO NOT PUT YOUR PHONE ON HOLD
- Please mute your phone line through web or phone controls
- please wait for facilitator to acknowledge the next speaker
- Identify yourself when speaking
What are the implementation date and scope of use for new model notices?
- The implementation date for the 8 new model notices and the service authorizations and appeals process changes is 4/1/18.
- The new model notices have been finalized for use by: Mainstream MMC, HIV SNP, HARP, MLTC Partial Capitation, Medicaid Advantage, and Medicaid Advantage Plus.
Can the model notices be modified?
- MMCP/HARP/HIV SNP may submit templates that modify the model notice for DOH approval. Instances where plans may need or choose to alter the model notices include, but are not limited to:
- Meeting systems limitations;
- Splitting the notices into multiple templates based on purpose of the notice;
- Editing placeholder language (while ensuring all content requirements are still met);
- Addition of a "to the provider" note for the right to reconsideration; and
- Making additions for NCQA requirements.
- MMCP/HARP/HIV SNP may elect to submit templates that do not follow the model notice for DOH approval; review may take longer.
Can previously approved Non–Discrimination and Language Services Templates be used for notice submissions?
- Plans may continue to use templates for non–discrimination and language services meeting 45 CFR 92 (§ 1557 of the ACA) requirements that have been approved by the Bureau of Program Implementation and Enrollment (BPIE) ––
- These must be updated to reflect large print content requirement as indicated in the models consistent with 42 CFR 438.10(d)(3) and (6).
- The plan contact information must be included in these sections (may not be a vendor contact).
Why is the inclusion of the service developer/manufacturer required in the FAD?
- Inclusion of the service developer/ manufacturer is a requirement specific to the FAD added by 10 NYCRR 98–2.9(e)(7). This information is to be included as applicable and available under the regulation.
- This should be the name of a company, not a restatement of what the item or service is.
- This information does not have to be included if the service developer/manufacturer is unknown to the plan.
What do the numbers on the Fair Hearing Request Forms mean?
- The codes included at the top of these request forms are the codes used by OTDA to process fair hearing requests.
- The codes indicate MLTC or MMC and whether or not the service is a home care service.
- Codes should be utilized as follows:
- General MMCP/HARP/HIV SNP= 229
- MMCP/HARP/HIV SNP Home Care Services= 266
- General MLTC= 212
- MLTC Home Care Services= 211
Is the plan required to include the carbon copy notation to the provider and enrollee representative?
- Plans are not required to keep the language regarding distribution of notices as included in the State approved models.
- If plans elect to alter this section of the notices, they must attest to and describe the process for appropriate distribution of the notices to these parties and document in their records that such notice was made.
How do new requirements and notices apply to retrospective claim denials?
- 42 CFR 438 does not change the timeframes for retrospective review determinations.
- The appeals and grievance system updates (i.e., exhaustion and new appeal/fair hearing request timeframes) apply to retrospective review.
- The model notices have been written to be used for prospective, concurrent, and retrospective review.
- Plans may elect to submit separate templates for different types of review.
What is the process for notice submission?
- Send templates to: MMCModelNotices@health.ny.gov
- Send complete submission:
- all parts of the template (letter, language/non–discrimination templates, and request forms)
- a signed coversheet for each template
- coversheet and template may be in the same e–file
- a single e–file should not include multiple templates or coversheets
- Unique identifier must be included on the coversheet and every page of the template
- Vendor templates:
- Each plan must submit its vendors´ templates
- templates received directly from vendors will not be reviewed
- vendor templates approved for one plan are NOT automatically approved for use by other plans
- Only plan reps can sign coversheets
- To expedite review, the unique identifier under which a vendor template has already been approved for use by another plan may be included in the comment box
- Each plan must submit its vendors´ templates
- Incomplete submissions will not be reviewed
What are the requirements for sending the enrollee´s case file?
- Enrollees may request and receive a copy of the case file or information from it at any time, free of charge
- The case file must be sent when an enrollee requests a Plan Appeal
- "sufficiently in advance of the resolution timeframe for appeals as specified in 438.408(b) and (c)"
- The case file includes: medical records, other documents/records, and any new or additional evidence considered, relied upon, or generated in connection with the Plan Appeal. This includes internally–generated documents.
- Case File is not the Evidence Packet
- The case file includes all information related to the review of the Service Authorization Request, Initial Adverse Determination, and/or Plan Appeal.
- The "evidence packet" contains information the plan will use to support the Final Adverse Determination at the fair hearing. The evidence packet must be sent to the enrollee when the plan receives notification of the fair hearing request from OAH.
Where did the new timeframe for filing a Fair Hearing come from?
- 42 CFR 438.408(f)(2) states the enrollee must request a fair hearing no later than 120 calendar days from the date of the plan´s notice of resolution.
- In the comments for the Final Rule, CMS provides that enrollees now have 120 days from the appeal resolution to request a fair hearing (see pages 27510, 27511, 27516).
When can an enrollee request Aid Continuing (AC)?
- The enrollee has the right to AC when:
- The plan makes a determination to terminate, suspend, or reduce a previously authorized service during the period for which the service was approved; or
- For an enrollee in receipt of long term services and support or nursing home services (short or long term), the plan makes a determination to partially approve, terminate, suspend, or reduce level or quantity of long term services and supports or a nursing home stay (long–term or short–term) for a subsequent authorization period of such services.
- In NYS, an enrollee is to automatically receive Aid Continuing (unless they specifically state they do not want AC) when the enrollee requests a Plan Appeal regarding these decisions within 10 days of when the plan sends the IAD or by the effective date of the IAD, whichever is later.
- Plans must immediately provide AC when so directed by OAH.
When does Aid Continuing stop?
The MMC plan must continue the enrollee´s services provided under AC until one of the following occurs:
- the enrollee withdraws the request for aid continuing, the plan appeal, or the fair hearing;
- the enrollee fails to request a fair hearing within 10 days of the plan´s written adverse appeal resolution notice (Final Adverse Determination) *;
- OAH determines that the Enrollee is not entitled to aid continuing;
- OAH completes the administrative process and/or issues a fair hearing decision adverse to the Enrollee; or the provider order has expired, except in the case of a home bound enrollee.
*Services authorized under AC must be continued for at least 10 days from when the Final Adverse Determination is sent.
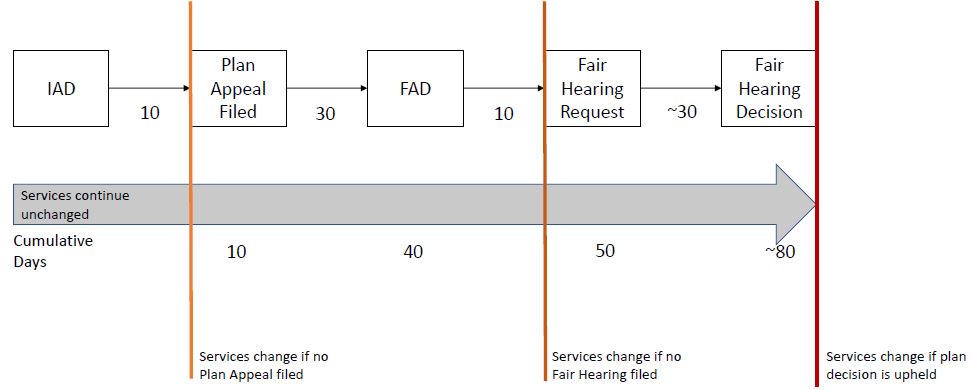
Aid Continuing – Standard Process
What should be included in the clinical rationale for decisions where specific criteria do not exist?
- For services without specific criteria, the plan may explain the medical practice standard or alternate clinical information that would demonstrate medical necessity.
- If there is not enough information to determine medical necessity:
- the criteria for approval must be included; and
- the plan must indicate how the information received was insufficient (or that no information was received) to make the determination; and
- the plan must state the specific information needed to make a medical necessity determination in the notice.
Updates to Specific Denial Reason/ Clinical Rationale Placeholders
INSERT IF THE DECISION IS CLINICAL AND ABOUT A REQUEST/CLAIM FOR A NEW SERVICE INCLUDING PARTIAL APPROVALS, AND IS NOT ABOUT LTSS, OR DELETE THIS SEGMENT
- You asked for [service] because [Insert the nature of the enrollee´s condition].
- To approve this service, {insert for partial approvals} [in full] the following criteria must be met: [Insert criteria required for the service to be approved].
- These criteria are not met because [Insert enrollee-specific details from the enrollee´s unique clinical/social profile to show why/how the enrollee does not meet the required criteria for service approval (necessitating a service denial) or why/how it does not fully meet the required criteria for service approval (necessitating a partial service approval) or Insert model prescriber prevails language or case–specific information about why the service is experimental/investigational.].
{Note: The clinical rationale must be sufficiently specific to enable the enrollee to determine the basis for appeal.}
Questions?
Thank You!
Please send comments or questions to: 438reg@health.ny.gov
Submit template notices to: MMCmodelnotices@health.ny.gov